Here’s an update on our 2016 article: Q & A with Migliore et al. about HITHOC and mesothelioma in Catania, Italy.
Migliore et. al at the University of Catania, Italy have just published the first randomized pilot study that directly compares hyperthermic intrathoracic chemotherapy with VATS pleurectomy / decortication with VATS talc pleurodesis. This is important because it represents a shift in the thinking surrounding treatment of Malignant Pleural Mesothelioma (MPM). For too long, too many surgeons have automatically shunted these patients into the palliative care treatment algorithm, which includes talc pleurodesis.
As we have discussed on multiple previous posts on this topic; treatments like talc pleurodesis are mainly performed for symptom relief. (The instillation of talc into the pleural space does nothing to treat the underlying cancer, but the talc pleurodesis slows the re-accumulation of pleural effusions which are a common cause of shortness of breath in these patients). HITHOC is different; it’s an active treatment aimed at treating the mesothelioma. A related treatment, called HIPEC (which is the same treatment aimed at cancers in the abdominal cavity) has rapidly become the standard of care for carcinomatosis, malignant peritoneal mesothelioma and other abdominally-based cancers.
Another important difference between this study and prior work in this area is the use of minimally invasive surgery for both groups. In several prior research studies, the use of large open operations in combination with HITHOC is believed to have contributed to an increased morbidity and mortality.
Multiple small studies (featured on this site) have shown increased survival and longevity for patients receiving HITHOC but these studies were not randomized. Randomization (while sometimes seeming to be cruel to enrolled patients) is important to eliminate conscious or unconscious treatment bias, and randomized control trials (RCT) are considered the highest level of evidence.
Why randomize?
Treatment bias is when researchers consciously or unconsciously select patients that they think will do better to place into one treatment group versus another. Sometimes this treatment bias is built into the study (ie. sicker patients enrolled into a palliative care arm of a study).
As you can imagine, if all of the high functioning, ambulatory, well-nourished patient with earlier stage cancers go into the treatment arm, and all the cachectic, bedridden patients with advanced cancer go into the other arm of the study, the results are more likely to favor the first group. Surprisingly, this sort of sorting strategy is not uncommon, and is sometimes used along with ‘non-inferiority’ trials to push expensive treatments and technologies. Migliore et al. lessen this by using patients at 3 separate study sites and randomizing them into two groups.
However, some selection bias will usually still exist, particularly when involved in a study in a specialized area like this – meaning that patients have to be referred to the study center in the first place. Hopefully, if the program is large enough and well-publicized in the local medical communities, referring physicians will send any and all of their patients with malignant pleural mesothelioma to be evaluated for enrollment. Once the researchers start receiving the referrals, then they use standardized inclusion criteria to enroll patients. This way, the patients selected are similar to each other, in cancer staging, functional status, age etc. Apples to Apples, so to speak.
How is a pilot study different from a ‘regular’ study?
As a pilot study, the main aim of the study was to recruit patients (to see if a larger future trial is practical or feasible). If you can’t get eligible patients into your studies, it doesn’t matter what medical breakthrough you might be working on.
This pilot study also have secondary goals; determining statistical significance (how many patients do we need to treat to show a statistically significant difference aka Number Needed to Treat (NNT), Survival rates at specific fixed intervals, length of stay, rate of peri and postoperative complications.
Who could participate (aka inclusion criteria)
In this particular study, all of the participants had to have a pleural effusion along performance status equal or below 2. This means that the patients had to be fairly functional and independent.
ECOG/WHO Performance Status (borrowed from verywellhealth.com)
0: Fully active, no restrictions on activities. A performance status of 0 means no restrictions in the sense that someone is able to do everything they were able to do prior to their diagnosis.
1: Unable to do strenuous activities, but able to carry out light housework and sedentary activities. This status basically means you can’t do heavy work but can do anything else.
2: Able to walk and manage self-care, but unable to work. Out of bed more than 50% of waking hours. In this category, people are usually unable to carry on any work activities, including light office work.
3: Confined to bed or a chair more than 50 percent of waking hours.Capable of limited self-care.
4: Completely disabled. Totally confined to a bed or chair. Unable to do any self-care.
5: Death
In addition to this, and patient participant consent, the participants had to agree to undergo VATS pleurodesis. (This last inclusion criteria may sound obvious, but if all your enrollees only agree to take the ‘experimental’ treatment, then the study isn’t random).
Patients with advanced disease, and patients who were too sick/ debilitated to undergo surgery/ anesthesia were excluded.
Potential limitations to randomization with this study design
In this pilot study, the randomization strategy is one of limited utility. In this study, recruited patients were ‘randomized’ based on which medical center they presented to. Now, that probably worked just fine when they were only recruiting 3 to 5 patients per year but this presents a potential problem for future, larger studies. Imagine, dear reader, after reading numerous articles here at thoracics.org, your loved one, family member, or even a neighbor is diagnosed with malignant pleural mesothelioma. Well, as an educated reader, and patient advocate, you are going to send your loved one to the treatment center that you know does the procedure you want. Depending on your oncologist, they might do the same. (We do it all the time in medicine when we refer patients to specific hospitals for “a higher level of care”/ surgical evaluation etc.). It wouldn’t take very long or very many patients for much of the medical community and the educated public to know patients enrolled in the trial at the University of Catania are in the treatment arm of the study, and getting HITHOC (Group B) and that the patients at Morgagni Hospital and University Hospital of Palermo (Group A) receive palliative treatment with talc pleurodesis. But given the relative scarcity of published information on HITHOC for the general public and in Italian, we can argue that for this small pilot study, this strategy worked. As long as the patients in the treatment groups look about the same, it shouldn’t affect the outcomes (that’s where performance status, and degree of disease comes in.)
Also, I would like to point out – that in this study, all of the patients continued to receive adjuvant therapy, which I think is really the only ethical option available. (If you know that talc pleurodesis is only of palliative value, it’s very questionable to require study participants to discontinue adjuvant chemotherapy, which may help slow the spread of their disease. We already know adjuvant chemotherapy doesn’t work that well, (hence the need for discovery of new treatments) but it seems almost punitive to make participants discontinue chemotherapy. So, while some many argue that this adjuvant treatment may impact results, the authors opted to take the more ethical route. Since everyone in the study was getting the adjuvant treatment, it can be factored into the study results.
As a pilot study, comparison groups are small. As we discussed before, one of the primary aims of this study was the recruitment of eligible patients – and it took several years (almost six) for the authors to recruit enough patients to be able to extrapolate data and publish this study. In this study, Group A had 14 people, group B had 13. As a pilot study, that is a respectable size (many pilot studies have groups in the single digits). However, this study size highlights one of the biggest limitations of pilot studies – and it’s also the reason that these authors don’t suggest changes to the treatment algorithm based on their results. Pilot studies are not designed to change treatment regimens – they are designed to see if there is enough of a reason to investigate further. (aka Is there something there? )
It’s just not enough people to make broad statements or changes to current treatment. The authors of this study acknowledge this.
A word about study size
Readers need to be careful to make sure they don’t fall into the trap of forgetting the importance of study and treatment group size. (This commonly occurs when the general media reports on medical findings. One of the best examples is the widespread reporting in the early 2000’s on the use of cinnamon as a treatment for diabetes. Millions of people at home adopted this as a more ‘holistic’ alternative, despite the fact that the preliminary studies had very few patients in the treatment (cinnamon arm). It wasn’t until 2013, that the first meta-analysis was published showing many of these claims to be misleading and exaggerated, and this meta-analysis was still based on multiple small size studies (see figure below)
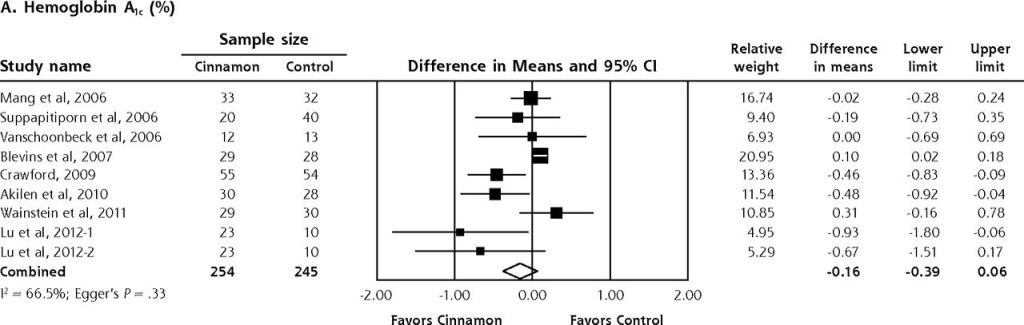
So now that we’ve discussed study size for this pilot study, let’s look at their findings and determine, Is there something there – an apparent difference in outcomes between the small groups important enough that a larger study should be conducted.
What were the actual treatments performed?
The patients in Group A had a talc pleurodesis via the Uniportal VATS approach that included a surgical biopsy for final diagnosis and tissue type.
Patients in group B underwent tissue biopsy prior to the procedure to confirm the diagnosis of Malignant Pleural Mesothelioma and tissue type. These patients then underwent pleurectomy / decortication via the VATS approach with mini-thoracotomy followed by the instillation of chemotherapy. The surgeons removed all of the parietal and visceral pleural as well as any visible tumor tissue (debulking). Then cisplatin, diluted with 2-3 liters of saline was heated to 41 degrees in temperature, and then circulated through the chest cavity for 60 minutes.
Results
Since I’ve included the link to the reference article, I am going to skip a lot of the discussion of group comparisons, (they were very similar), hospital stay (very similar) and the rates of post-operative complications were very similar (group A 8 patients, group B 7 patients).
Let’s look at the big question for the participants in the study and their families – and the real reason Dr. Migliore and all of his colleagues are investigating HITHOC as treatment.
Patient survival
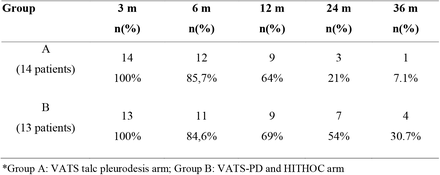
Look at the last column – at 36 months (3 year survival):
4 patients in the HITHOC treatment group were still alive versus just one in the talc pleurodesis cohort. The authors note that this survival for the HITHOC group might even be skewed a little, in that some of the patients in the HITHOC group didn’t receive treatment until SIX months after diagnosis (and all survival rates are calculated as length of survival after diagnosis).
So, yes, even with these small, small numbers, these findings are important enough for researchers to continue investigate in this area. It certainly warrants a larger study, research grants/ and other financial support.
However, it also needs to be noted, that researchers in this study found that the tumor tissue type had a major impact on outcomes. Patients with epithelioid MPM lived on average of 15 months after talc pleurodesis ( 9 patients) versus 45 months after HITHOC ( 9 patients). Patients with biphasic tumor type, or sarcomatoid type were less frequent in this study, but it appears to carry a poorer prognosis.
Reference article:
Migliore, M. et. al. (2021). Comparison of VATS Pleurectomy/Decortication Surgery plus Hyperthermic Intrathoracic Chemotherapy with VATS talc pleurodesis for the treatment of Malignant Pleural Mesothelioma: a randomized pilot study. MedRxIV, 28 Nov 2021.
For more about HITHOC, please see our archives.