We are here with the latest HITHOC pilot study updates from Dr. Migliore et al. If you remember, Dr. Migliore and his colleagues in Sicily have been investigating and researching the effectiveness of HITHOC for well over a decade. In fact, we’ve talked about this study before, when we presented preliminary findings. But now the authors are presenting the results of a six-year pilot study with long-term follow-up in a paper that was published in the scientific journal, Cell. (Original paper here).
Why this is important
In the general population, patients diagnosed with malignant pleural mesothelioma (MPM) have a mean survival of 9 to 12 months.
The mean age at time of death was 70, meaning this disease affects relatively young patients after a long incubation period. It affects men at a ratio of almost 4 men (3.6) to one woman. (Presumably, this is related to occupational/ industrial exposures and trends – few women work/ worked in shipyards, construction).
While many people think that asbestos related mesothelioma is a thing of the past (or time limited due to the fact it has known carcinogenic effects), it is still being used in many products in several countries including the United States.
How do we treat Pleural Mesothelioma now?
Despite this, the standard of treatment is generally palliative in nature. Talc Pleurodesis is used to drain existing fluid around the lung, and then the talc is used as a sclerosing (or scarring) agent to prevent the pleural surfaces from being able to secrete more fluid. This makes the patient feel better because they breathe better – which is a very important consideration for patient care – but does nothing to treat the underlying cancer or prevent its spread. Patients generally live around 14 months after this procedure, when it is performed for mesothelioma.
Other surgical treatments have been tried in the past including Extrapleural pneumonectomy (EPP), which has a high mortality rate, and Pleurectomy/ Decortication.
Pleurectomy / Decortication, which has replaced EPP in many cases, has a low surgical mortality (1.8%), but carries a high risk of recurrence, meaning many patients’ cancer will return. The average survival for patients having this procedure is only 17 months.
In contrast, there have been several small studies that suggest that HITHOC offers greater survival for patients with mesotheliomas – with the average survival of 20 to 35 months. But these studies have been small, and many thoracic surgeons and oncologists remain unaware of HITHOC as a potential treatment option. Others remain skeptical of its potential benefits (which is not necessarily a bad thing!) So, Dr. Migliore and his colleagues designed this study to see if a larger scale trial with more participants would be feasible or worthwhile.
Pilot studies like this one are used to determine how many patients need to be enrolled to see a statistically meaningful result, and if there is a “meaningful” result at all.
This is important because many of the studies mentioned in general media do not meet this criteria but are widely reported as successful nonetheless just because of so-called ‘newsworthiness’. Every time readers see a story on a seemingly miraculous cure based on a garden variety supplement for a wide host of medical problems (depression, arthritis, heart disease, Alzheimer’s or you name it), this is an example why pilot studies are needed to both protect the public and to advance medicine using scientific evidence. (We’ve talked about this before.)
So what does this pilot study show?
As you may recall, the preliminary results from this study, (as we reported in 2021) showed favorable results in the HITHOC treatment group, with improved survival rates, despite the fact that many of the patients that were randomized into the HITHOC treatment group actually had more advanced cancers.
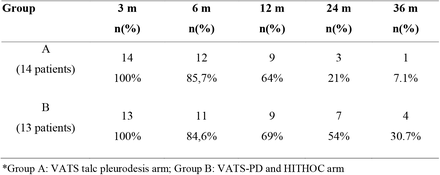
The final data confirms this with 30% of the HITHOC patients (4 out of 13) alive at median follow up of 28 months. (Compared to the pleurodesis group which only had one patient (out of 14) still alive at 19 months. For the full results of this trial – please see the original article.
The biggest limitation for the researchers in this trial was recruiting patients for enrollment. They reached out to multiple medical centers and oncology practices – and received referrals for only five patients a year, despite the fact that Biancavilla, in Sicily is one of the mesothelioma hotspots. This small number met the threshold for statistical significance for a pilot study – but falls far short of what would be needed for a larger, more powerful study.
but for large scale, multi-site international trials in the future, we need to do a better job at connecting eligible patients with research studies..
Now what? What’s next ?
Now as we wait to see if other surgeons and oncologists will answer the challenge – and participate in a larger, multi-center (international) randomized control trial with hundreds, if not thousands of patients, to compare HITHOC to other treatments. When, and if, that happens, I will report enrollment information for potential referring physicians and patients.
Other news in the treatment of MPM
In the meantime, we await further results from the MARS2 trial, headed by the dynamic Dr. Eric Lim, in the UK. These results will be presented in Singapore, this fall.
The MARS2 trial, unfortunately, doesn’t use any HITHOC protocols but looks at whether pleurectomy/ decortication in addition to chemotherapy alone enhances survival and quality of life. Now, if only Dr. Lim would introduce some of that chemotherapy (preferably Cisplatin) at 42.5 degrees into the chest cavity. Then we’d really have something to talk about!
References/ additional information
Featured article: Migliore et al. (2023). Comparison of video-assisted pleurectomy /decortication surgery plus hyperthermic intra-thoracic chemotherapy with VATS pleurodesis for treatment of Malignant Pleural Mesothelioma: a pilot study. Cell, 25 May 2023.
Marinaccio et al. (2015). Malignant mesothelioma due to non-occupational asbestos exposure from the Italian national surveillance system (ReNaM): epidemiology and public health issues. Occup Environ Med 2015 Sep;72(9):648-55. doi: 10.1136/oemed-2014-102297. Epub 2015 Jun 4.
Nuyts, et al. (2018) Hotspots of Malignant Pleural Mesothelioma in Western Europe. Transl Lung Cancer Res. 2018 Oct; 7(5): 516–519.
As we’ve talked about in a previous interview with Dr. Isik, (Turkiye) there are regional areas where natural conditions (such as asbestos in the soil) lead to mesothelioma clusters. There are also areas, like the naval shipyards in the United States, where occupational/ industrial exposure leads to disease clusters. In this investigation of mesothelioma clusters in Western Europe, the majority were industrial/ occupational in nature.
Is asbestos illegal?, The Mesothelioma Center, consumer information by lawyers for mesothelioma patients.
Banning Asbestos, Mesothelioma.com, consumer information by lawyers for mesothelioma patients.
Palliative versus curative care in Mesothelioma Mesotheliomafund.com – another legal site for mesothelioma patients.
HITHOC in Germany, a tale of two cities. Thoracics.org